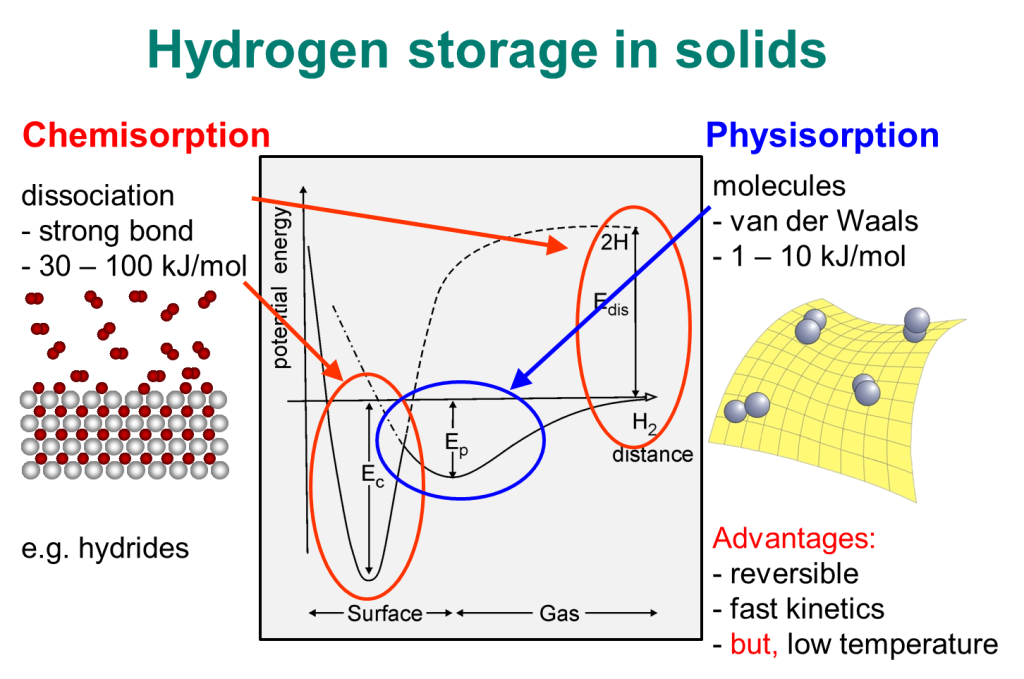
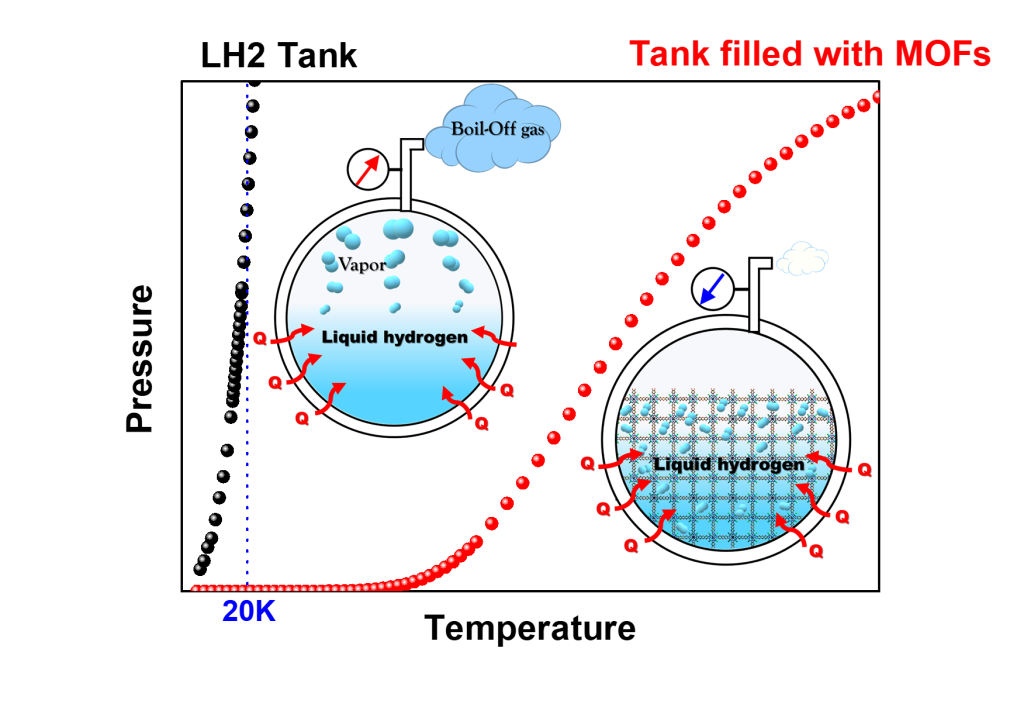
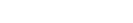Our Lab at UNIST researches molecular physisorption in nanoporous materials in cryogenic conditions. We exploit such phenomena to solve industrial challenges, such as
1) Efficient energy carrier storage(e.g., hydrogen, methane, etc.),
2) Greenhouse gas capture (e.g., CO2, NH3) ,
3) Quantum Sieving for cost-effective light gas isotope (e.g., H2/D2/T2, 16O/18O, 3He/4He, etc.) separation
4) Novel H2 boil-off mitigation technology for liquid hydrogen storage and transportation,
5) Magnetocaloric effect(MCE) of MOF for LH2 production.
In particular, efficient hydrogen storage technology will become crucial for the hydrogen economy to achieve carbon neutrality in the future. However, there are still many technical difficulties to be solved, and the main research direction of our team is to solve them. In addition, isotope separation technology using nanoporous materials is spotlighted as a new technology that may replace the expensive cryogenic distillation method. And we are the leading group in these related fields. Moreover, the industrial demand (e.g., semiconductor and display fields) for deuterium is rapidly increasing. Tritium separation and removal technology are also heavily required in nuclear fission and fusion reactors. Therefore, these isotopes separation technology will receive a lot of attention in the near future.
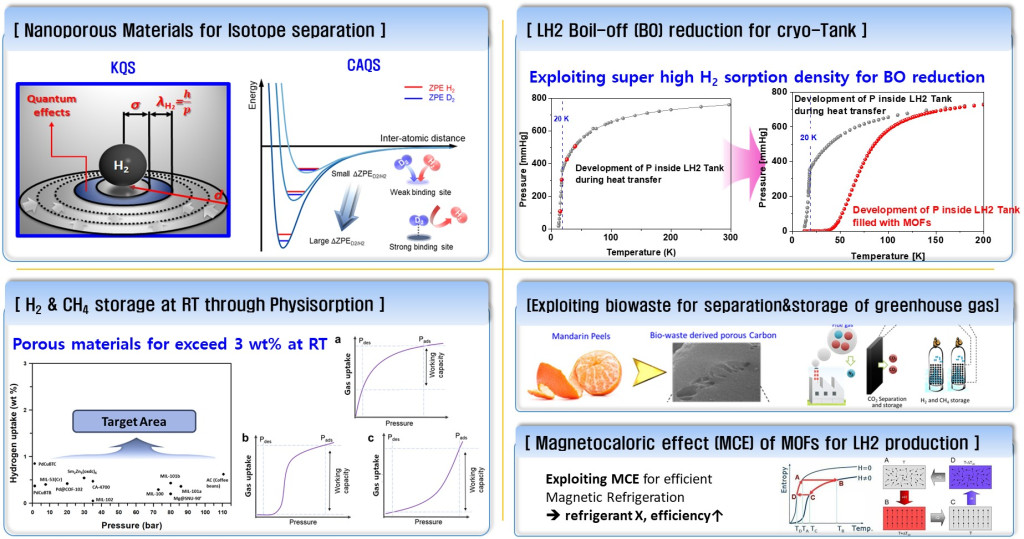
Research area
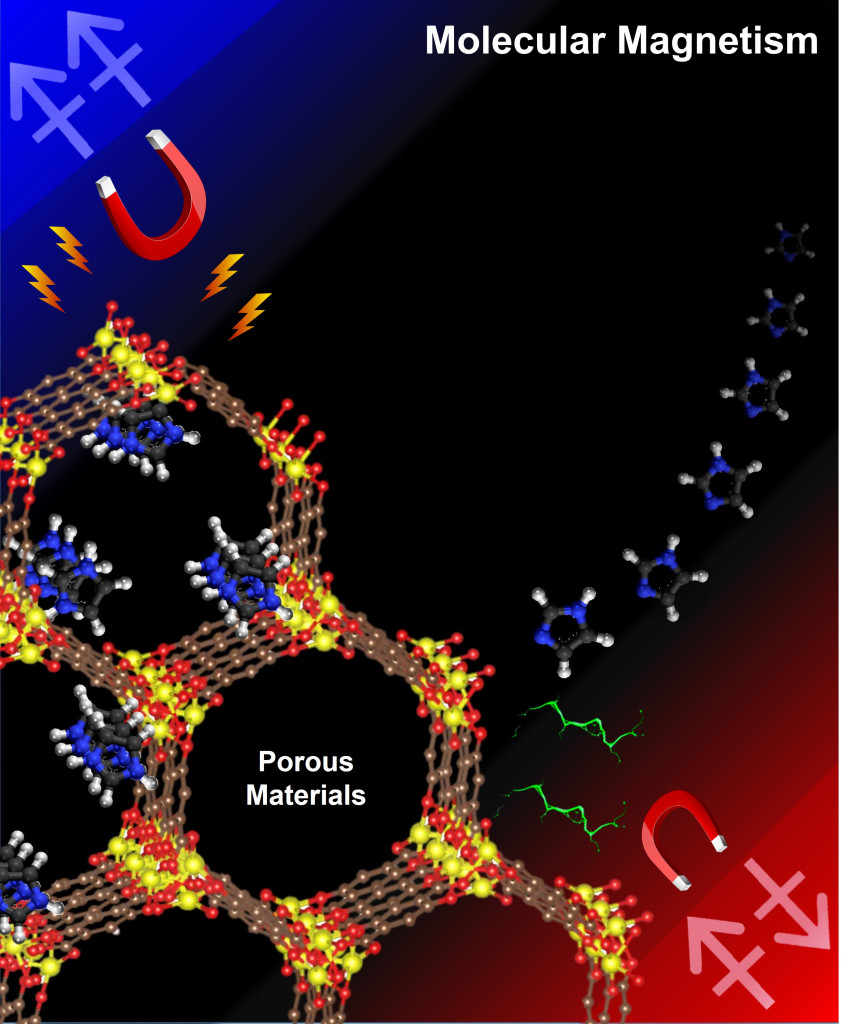
The magnetic properties in metal−organic frameworks (MOFs) are experimentally and theoretically investigated. The magnetically ordered state at low temperatures can be altered via guest molecule coordination. Guest molecule coordination influences the spin state of Metal ions with orbital geometries, resulting in observed shifts in the critical temperature and field. The magnetic behavior in MOFs reveals the coexistence of spin canting, metamagnetism, and magnetic phase transition.
Molecule-based magnetic materials have recently attracted increasing interest for use in novel applications (e.g., magnetic switchers, magnetic sensors, and multifunction magnetic devices in molecular spintronics) because of their ability to display ferromagnetism and other more complex magnetic phenomena. In particular, these materials are advantageous because their magnetic properties can be controlled by changing their spin center, the length of their interactive organic ligands, or both. These advantages permit the study of important magnetic features such as magnetic dynamics and magnetic hysteresis with high spontaneous magnetization. Various magnetic systems with designed novel structures in single-molecule magnets (SMMs) and single-chain magnets (SCMs) have been reported.
Among the various types of molecular-based magnets, metal–organic frameworks (MOFs) could represent a potential model system because of their numerous metal clusters within a microporous crystalline structure, their varied functionality (via ligand and metal cluster exchange), and/or their controllable cluster–cluster interactions through tunable pore size and structure. Moreover, open-metal sites (OMSs; unpaired electrons in the metal cluster) in MOFs are very useful for understanding the influence of unpaired electrons on magnetic metal ions, by the design of interior structures in the frameworks. For example, the antiferro- and ferromagnetic (AFM and FM, respectively) coupling effects on metal spin centers in OMSs can therefore be elucidated via the gradual occupation of unpaired electrons at OMSs. Moreover, the spin canting and metamagnetism are particular types of AFM coupling effects in some low-dimensional [layers, two-dimensional (2D); chains, one-dimensional (1D)] systems. Because the metamagnetism disrupts the AFM ordering and turns into the FM state when a certain external field is applied, FM and canted AFM couplings should be coexist in interchain interactions with intrachain interactions.